
WebDraw a pyranose ring containing five carbon atoms and one oxygen atom. at each chiral carbon as R or S in this way carbons 2 through 5 in dextrose pt gac samudera logistics alamat how many asymmetric carbon atoms are present in ribose answer 5.4: Stereogenic Centers - Chemistry LibreTexts 6.1: Structure and Function- Carbohydrates Solved a, Draw the structure of the ring form of D-ribose. Glucose has four chiral carbons in its aldehyde form, and so there are 24, or These 16 isomers are shown in Fig. Sucrose, with nine chiral carbons, has 29stereoisomers, or 512. ptg advies amersfoort how many asymmetric carbon atoms are present in ribose answer What is an Asymmetric Carbon? - Definition, … 3.5: Carbohydrates - Biology LibreTexts Webin a molecule there are as many as 2npossible stereoisomers. The configuration of such a molecular unit is chiral, and the structure may exist in either a right-handed configuration or a left-handed configuration (one the mirror image of the other). ptg acl surgery WebA carbon atom that is bonded to four different atoms or groups loses all symmetry, and is often referred to as an asymmetric carbon. 
An asymmetric carbon atom is defined as a carbon within an organic compound that contains four different atoms or groups of atoms (substituents) bonded to it.Since there are three chiral centers in this constitution, … Web 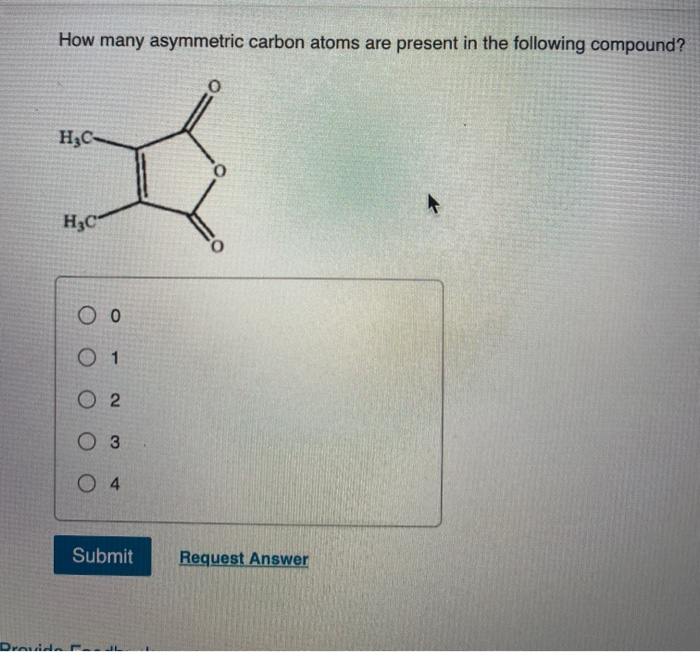
pt gag nikel how many asymmetric carbon atoms are present in ribose answer WebThere are eight stereoisomers of 2,3,4,5-tetrahydroxypentanal, a group of compounds referred to as the aldopentoses. What is the value of n in 2,6-dimethylnonane? How many. The base units in DNA are … WebHow many asymmetric carbons are present in a-D-ribopyranose? A) 0 B) 1 C) 2 D) 3 E) 4 The general formula for alkanes is C_nH_. How many asymmetric carbons are present in each?. Aidopentose has three asymmetric carbon atoms whereas aldohexose has four asymmetric carbon atoms. The laboratory stuff that was tested contained only one stereo-isomer, but the industrial product also contained the other one, which led to great deformities in newborn babies.How many asymmetric carbon atoms are present in ribose Ribose - Chemistry LibreTexts 25.4: Configurations of Aldoses - Chemistry LibreTexts Aldose - Wikipedia WebAidopentose has three asymmetric carbon atoms whereas aldohexose has four asymmetric carbon atoms. It will be secreted by the kidneys, where it may do some damage in the long term, or part of it may cause fermentation in the intestines (flatulence).Ī more notorious example is thalidomide (aka softenon and other names), prescribed in the 50s and early 60s for pregnant women to deal with morning sickness (a.o.). It tasts just as sweet, but no calories can be gained from it. It is possible to make L-glucose (its mirror-image) by chemical synthesis. One example of a chiral molecule is glucose, that naturally only occurs in the so-called right-hand variety, called D-glucose or dextrose (dexter=Latin for right). Most organisms are 'keyed' to processing only one of these forms, and that is the form that usually occurs as the only one in nature, because they are also produced by (other) organisms.Ĭhemical synthesis of compounds very often produces an mixture of both forms, called a racemic mixture, where only one form is effective, and the other form is not used or may even do damage. The simplest form are mirror images of each other, like a pair of gloves. Whenever there are asymmetric atoms in a molecule, there is the possibility of stereo-isomers. Because organisms may react differently to stereo-isomers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
